Lithium hexafluorophosphate has always been the main electrolyte lithium salt used in commercial lithium-ion batteries. The main reason why it still dominates the commercial lithium-ion battery electrolyte lithium salt market is that it can form an appropriate solid electrolyte phase interface film on the carbon anode and effectively passivate the Al current collector. Lithium hexafluorophosphate, as the lithium salt of lithium-ion battery electrolyte, in addition to the purity of the product, mainly requires that the product contains less water and free acid (calculated as HF). Under normal circumstances, it should at least have the following quality indicators:
Purity greater than 99.9%;
(2) The content of H2O is less than 20 PPM;
(3) The HF content is less than 80ppm.
LiPF6 is usually produced using phosphorus halides and LiF or LiCl as raw materials, with anhydrous hydrogen fluoride as the solvent. The crystallization separation of products produced by this process is relatively easy and it is easy to achieve industrialization. It is currently a relatively mature production process route for LiPF6. However, how to remove water and hydrogen fluoride from the crude LiPF6 product to meet the requirements of the lithium-ion battery industry is one of the key steps in the production of LiPF6. However, LiPF6 is highly hydrolyzed and has poor thermal stability, which makes the purification of LiPF6 extremely difficult.
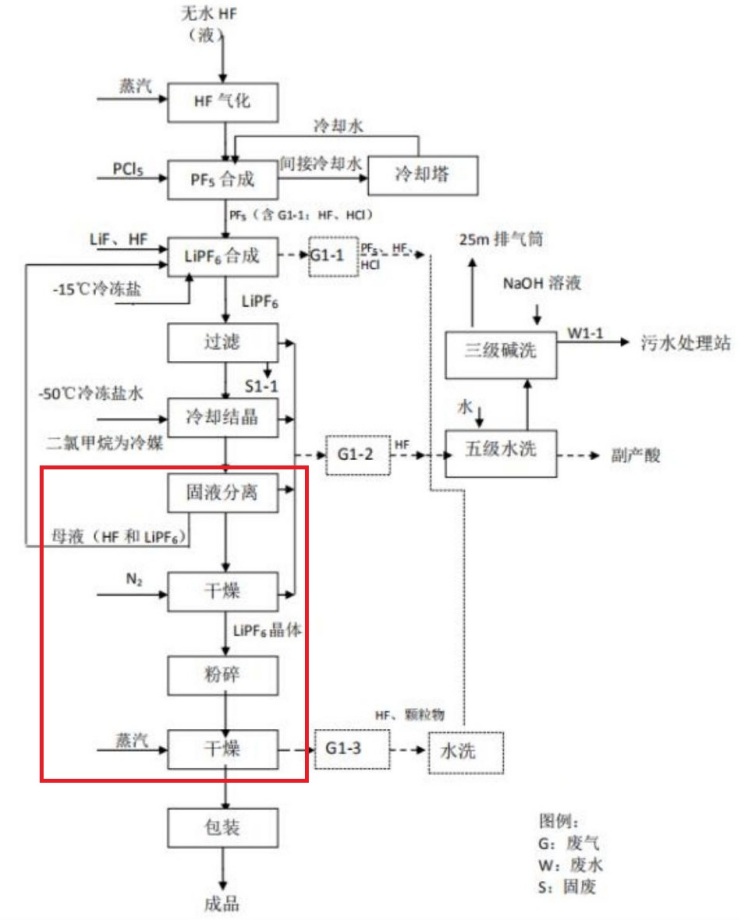
Analysis of the current filtration and Drying Process of Lithium hexafluorophosphate
(1) Filtration: After the synthesis is completed, the liquid is filtered to remove a small amount of insoluble impurities (mainly phosphate ions formed by a small amount of calcium and iron ions brought in by lithium fluoride), and then enters the synthesis liquid storage tank. The filtration is natural gravity filtration. Then, the synthesis liquid is transported to the crystallization tank through stainless steel pipes by a pump. This process generates filter residue. A small amount of HF that overflows from the filtrate enters the exhaust gas.
(2) Cooling, crystallization - liquid release
Crystallization is carried out by static crystallization, with lithium hexafluorophosphate cooled and crystallized at -20℃. After the crystallization process is completed, solid-liquid separation is carried out. The mother liquor after separation is sent to the mother liquor storage tank for recycling. A small amount of HF that overflows during the cooling, crystallization and liquid discharge processes enters the exhaust gas.
(3) Mother liquor separation and pre-drying (double-cone dryer with filtration function)
The mother liquor enters the double-cone dryer with filtration function through a closed pipeline from the crystallization tank. Nitrogen is introduced to create positive pressure, causing solid-liquid separation, and then the initial drying is carried out.
(4) Crushing of agglomerated materials (crusher
The initially dried lithium hexafluorophosphate crystals are sent to the mobile container holding the crystals through a primary classifier. The lithium hexafluorophosphate crystals in the mobile container are protected by nitrogen seals and then enter the crusher to be crushed to the target particle size.
(5) Secondary drying (double-cone dryer or paddle dryer)
The crushed lithium hexafluorophosphate crystals are directly fed into the dryer and heated and dried with 85℃ hot water (jacket) under nitrogen seal protection. A small amount of fine powder carried out by nitrogen during the drying process is collected by a fine powder collector and reused, while the uncollected part enters the exhaust gas.
(6) Graded packaging
The lithium hexafluorophosphate crystals dried under nitrogen seal protection enter the vibrating classification screen, and the qualified products are packaged under nitrogen seal protection.
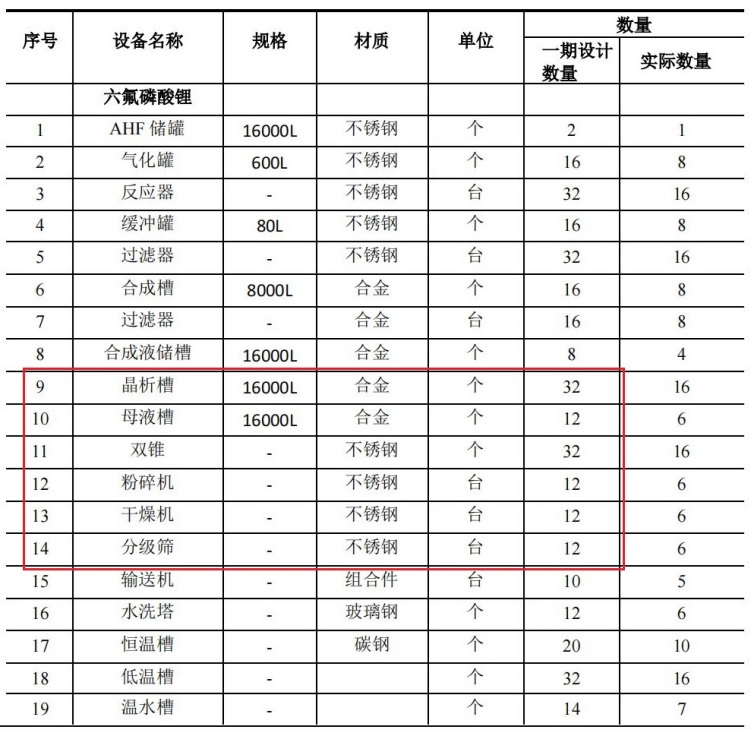
List of current filtration and drying process equipment for an annual output of 5,000 tons of lithium hexafluorophosphate
There are problems with the current filtration and drying process of lithium hexafluorophosphate
(1) Conventional refining processes require filters, crushers, dryers, etc. Each of them has its own supporting equipment, which is costly and occupies a large area.
(2) There are many processes and equipment, the equipment structure is simple, the operation is complicated, and the labor intensity of production is extremely high.
(3) The entire set of equipment cannot be operated normally or is subject to high labor intensity, resulting in a long production cycle, high energy consumption and low efficiency, which affects product quality.
(4) Material transfer is difficult, the labor intensity is high, and it is also prone to material loss and waste.
(5) Materials need to be transferred multiple times, which is prone to cross-contamination and makes it difficult to meet the requirements of full sealing.
(6) For flammable, explosive, toxic and highly toxic materials, there are potential safety hazards.








 备案号:
备案号: