The common production process of crystalline and dried active pharmaceutical ingredients is as follows: crystallization → filtration → washing → filtration → drying → discharge → crushing → sieving → powder mixing → packaging. The traditional and backward process flow is manifested as follows: crystallization in a crystallization tank → solid-liquid separation by a centrifuge (or solid-liquid separation by a filter)→ cleaning in a washing tank → vacuum filtration → drying in a double-cone rotary vacuum dryer (or vacuum drying oven)→ manual discharge → crushing by a crusher → screening by a mechanical vibrating screen → mixing by a mixer → manual loading into cylinders. In this process and equipment operation, there were multiple transfers of crystal-containing materials, and the continued use of traditional equipment was subject to certain limitations. The problems exposed by such traditional crystalline drying type raw material drug production equipment and technology are:
In the production of crystal-containing materials, there will be multiple processes such as shoveling and turning the material, scooping out the powder, and pressing and grinding the filter cake. This will cause the crystals to be exposed for too long and also lead to cracks in the filter cake, affecting the uniformity of crystal formation and thus the quality of the product.
(2) Due to the limitations of using traditional equipment, materials need to be transferred multiple times during production, which is prone to cross-contamination and makes it difficult to meet GMP requirements.
(3) The numerous processes and equipment, along with the simple structure of the equipment, make the operation cumbersome and the labor intensity of production extremely high. Meanwhile, the backwardness of traditional individual equipment makes the cleaning and sterilization of batch change extremely difficult, and it is impossible to ensure its effectiveness.
In conclusion, the biggest problem exposed by traditional crystalline drying type raw material drug production equipment and technology is the difficulty in meeting GMP requirements.

Wuxi Shuangrui Machinery has integrated the traditional crystallization and drying process of active pharmaceutical ingredients, making breakthroughs on the basis of inheriting the essence of tradition. Starting from equipment reform, integrate its processes to form a new overall process equipment solution for the crystallization and drying of active pharmaceutical ingredients. Starting from simplifying the process, the filtration, washing and drying processes are combined on one machine to complete, reducing the number of process transfers. Starting from automation, washability and sterilization, improvements should be made to make the overall process more suitable for the production of sterile active pharmaceutical ingredients. The improved process equipment scheme for the API active pharmaceutical ingredient (API) fine drying and packaging production line is as follows: crystallization in the crystallization tank → filtration, multiple washing and filtration, drying and discharging by the "three-in-one" dryer → crushing by the specially developed crusher → screening by the new type of vibrating screen → mixing by the mixer → loading into the cylinder by the automatic filling and weighing machine. Among them, most processes can be transferred and transitioned through a closed material transfer system.

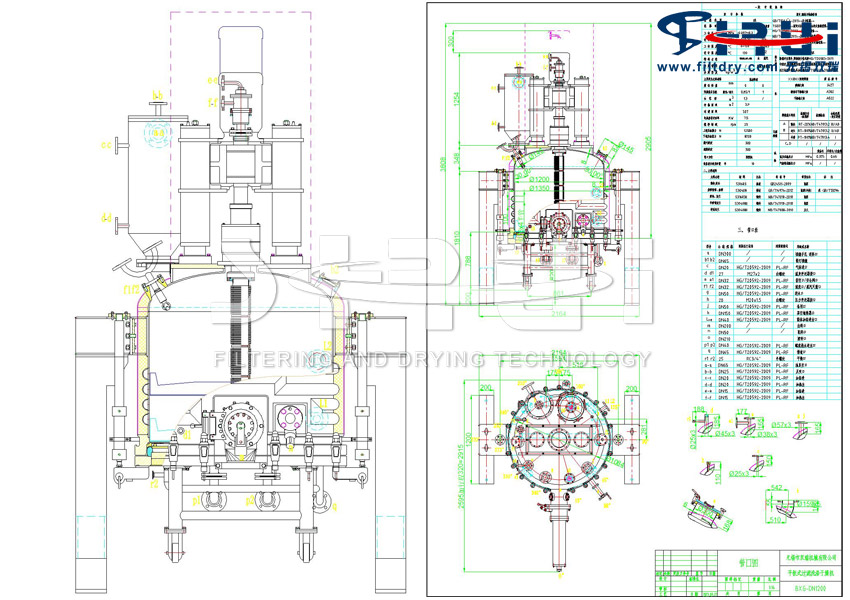
Introduction to the Structure of the Three-in-One Drying Machine for API Active Pharmaceutical Ingredients
The API raw material medicine precision drying and packaging three-in-one dryer mainly consists of a tank, a base, a drive device, a stirring device, a heating device, a side discharge valve, a support, etc.
(1) Tank body: It is a sealed container with an elliptical head at the top and a flat bottom at the bottom. If drying is required, a jacket or half-tube heating can be used on the cylinder wall. The material of the container is selected based on the properties of the material and the process conditions.
(2) Chassis: It is divided into two types: fixed type and detachable type.
It is composed of a supporting structure, a heating device, a filtering medium, etc.
I. The fixed chassis is welded to the equipment cylinder and cannot be disassembled. It is generally applicable to the filtration of materials that do not clog or are easy to clean.
Ii. The detachable chassis is composed of the chassis and the tank independently, connected by hook-shaped bolts. The hydraulic cylinder is used to lift and lower the chassis, thus enabling its installation and disassembly. The lower part of the chassis is equipped with four universal wheels, which facilitate the removal and cleaning of the filter plates.
(3) Filter medium: Generally, filter cloth, metal mesh or sintered plate is used.
(4) Drive unit: Located at the top of the head. There are two types of transmission methods: motor-reducer and hydraulic motor. The rotational speed can be adjusted.
(5) Stirring device: Composed of sealing components, stirring shafts, agitators, etc. The stirring device can achieve variable speed, clockwise and counterclockwise rotational movements, as well as constant-speed upward and downward movements.
According to the form of the stirring blades, they can be classified into straight blade blades and S-shaped blade blades. According to requirements, the cavity inside the blade can be heated by heating medium through the rotary joint at the shaft end to heat the material. The forward rotation achieves the functions of stirring and mixing as well as automatic discharge, while the reverse rotation helps to smooth the surface of the filter cake.
(6) Heating device: The heating of the fully enclosed four-in-one crystallization, filtration, washing and drying integrated machine has the following heating methods:
The cylinder is equipped with half tubes or jackets, which are the main heating components provided.
Ii. Heating device inside the chassis.
Iii. Hollow blades connected through the rotary joint at the shaft end.
These heating structures can be used in combination or individually.
(7) Lateral discharge valve: It is divided into manual and hydraulic types according to the operation form.
After the valve core is closed, the inner side of the valve body fits with the inner wall of the equipment, which will not affect the stirring on the inner side. The seal of the discharge valve can be either a soft seal or a hard seal. For products with aseptic requirements, an online sterilization device and an online cleaning device can be added inside the discharge valve.
For general chemical requirements, a quick-opening manhole device can meet the requirements.
(8) Support: It is a vertical support, made of welded steel structure.
The process flow of the three-in-one drying machine for API raw material medicine precision drying and packaging
The API API raw material medicine precision drying and packaging three-in-one dryer is a multi-functional device that can successively complete multiple unit operations such as stirring, reaction, filtration, washing, deliquid removal, and drying in one container. It has the advantages of simplifying the process, improving production efficiency, preventing material contamination, steam sterilization, convenient material replacement, and mechanical and electrical integration. At the same time, it can significantly reduce the area of clean workshops throughout the entire product production process, greatly lowering the basic investment cost for customers. It is widely used in the pharmaceutical, pesticide, biochemical, food, beverage, fine chemical, dye and other industries, and is particularly suitable for production occasions with high cleanliness requirements and small batch and multiple varieties.
(1) Crystallization: Crystallization is an important process operation in the production of sterile active pharmaceutical ingredients. The quality of drug crystal formation not only has a significant impact on the difficulty of subsequent filtration, drying, crushing and packaging operations, but also plays a crucial and decisive role in key quality indicators such as the color grade, dissolution time and stability of the drug.
(2) Filtration: Solid-liquid separation is achieved under pressure or vacuum conditions. After filtration, the filter cake is flat and the solid-liquid separation effect is good.
(3) Washing: A universal cleaning ball is installed on the inner side of the container to evenly distribute the cleaning solution within the container, which is used for material cleaning and container cleaning. Through the stirring of the lifting and lowering stirring blades, the filter cake and the cleaning solution are mixed to thoroughly wash the slurry suspension filter cake.
(4) Drying: The filter cake is gradually loosened layer by layer by the stirring blades. The heating device of the equipment heats the filter cake evenly. Under vacuum conditions, hot gas is used to heat the wet material to accelerate evaporation and achieve the drying purpose.
(5) Unloading: After the material is loosened by scraping, it can be automatically discharged from the discharge port on the side of the cylinder wall by pushing the material through the stirring blades.
(6) The entire set of equipment features simplified processes, prevention of cross-contamination, convenient material interchangeability, enhanced production efficiency, and integration of mechanics and electronics. It can adopt PLC control technology to achieve fully automated operation throughout the filtration and drying processes.
(7) The entire operation process is completed in a closed, aseptic clean working area, avoiding direct contact between materials and people as well as contamination by the surrounding environment.
The application scope of the API raw material medicine precision drying bag three-in-one dryer
Solid material recovery such as pesticides, medicines, chemicals, dyes, solvent recovery, washing required, flammable and explosive, highly toxic occasions; The fully enclosed four-in-one crystallization, filtration, washing and drying machine is applicable in situations where washing is required, such as diphosphorescence, glyphosate, pyrethrin pesticides, vitamins, penicillin, etc. The four-in-one filter is mainly used in situations where washing, drying, and the recovery of solvents (such as toluene, methanol, ethanol, ethyl acetate, cyclohexane, etc.) are required.
The problems existing in the traditional filtration, washing and drying processes of active pharmaceutical ingredients include poor washing effect of filter cakes, high labor intensity of manual operation, difficulty in material transfer, easy influence and contamination of materials by equipment transmission components and open environments, and difficulty in operation and verification of equipment cleaning and sterilization, etc. These serious defects have a significant impact on the color grade, contamination and visible foreign matter quality indicators of the drugs.
In recent years, the fully enclosed four-in-one crystallization, filtration, washing and drying integrated machine developed by Shuangrui Machinery has been rapidly promoted and applied in the production of raw material medicine filtration and drying due to its superior performance of being able to complete filtration, washing, drying, sampling, automatic discharging, as well as cleaning and sterilization in the same container. It has gradually replaced traditional crystallization and drying production equipment such as centrifuges, vacuum filtration tanks, box-type vacuum dryers and double-cone rotary vacuum dryers, and has become a new type of equipment for the API raw material drug fine drying and packaging production line.




















 备案号:
备案号: