Valsartan is a new generation of angiotensin II receptor antagonist, featuring high selectivity and specific direct effects. It can effectively antagonize the AT1 receptor, with a half-life of approximately 9 hours. Its antihypertensive effect can be maintained for more than 24 hours, and it has a good trach-to-peak ratio. It has a wide range of therapeutic effects and is well tolerated by patients. The general dosage of valsartan is 80 milligrams per day. It can effectively lower blood pressure for patients with mild to moderate hypertension.

As valsartan is a chronic disease medication, patients need to take it for a long time. To prevent side effects caused by the accumulation of impurities in the product in the patient's body, the 2010 edition of the Chinese Pharmacopoeia stipulates that the total impurity content in the raw material of valsartan should not exceed 0.30%, and the content of a single unknown impurity should not exceed 0.10%. The impurity content in the active pharmaceutical ingredient of valsartan is greatly influenced by the quality of the raw materials used in its preparation process, especially by the quality of the raw material n-valproyl chloride. The detection of the raw material n-valproyl chloride usually requires its derivatization and then chromatography, which often leads to the impurities not being completely and effectively detected, and thus may be transferred to the final product. Once the impurity content of valsartan exceeds the limit stipulated by the quality standard, it is required to be purified. Therefore, it becomes crucial to determine an economical and effective method for purifying valsartan.
The structure of valsartan has A chiral center. During the refining and purification process, when valsartan is heated and dissolved in a solvent, its chiral center is prone to racemization, generating D-type valsartan isomers (EP pharmacopoeia impurity A), which leads to a decrease in the optical purity of the refined valsartan product. At present, most of the existing patent reports on the removal of D-type valsartan isomers use simple ester solvents or mixed alcohol ester solvents for repeated refining of valsartan. The disadvantage of using this refining method is that after production scaling up, the D-type valsartan isomer of valsartan continuously increases with the extension of the refining, purification and drying process time, which is unfavorable for the removal of D-type valsartan isomers. Moreover, the drying time is long and the residual solvent is relatively high, which is not suitable for the industrial production of high-standard valsartan.

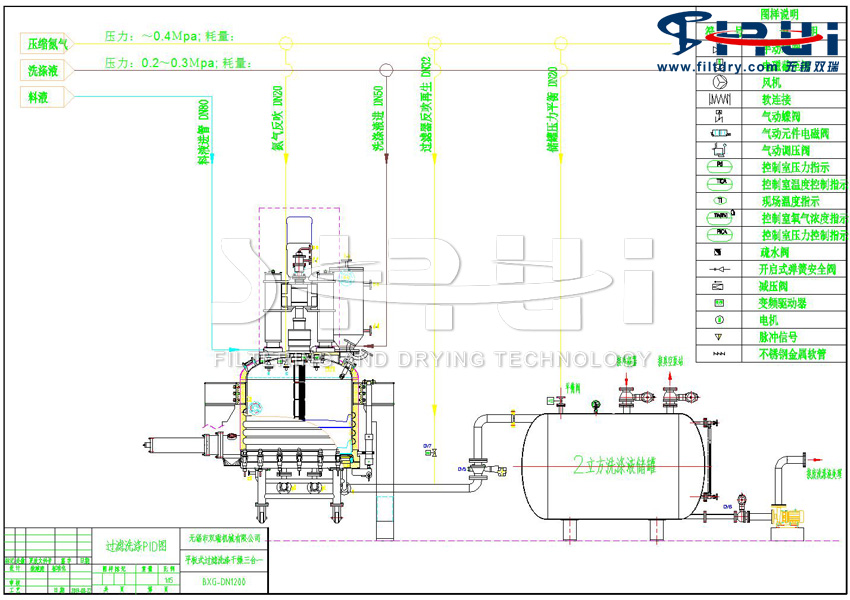
The working principle of the three-in-one filtration, washing and drying system for the refined antihypertensive raw material drug valsartan, which is of pharmaceutical grade
The common purification method for the antihypertensive raw material valsartan shows significant racemization during the cycination process, and there is no separation and purification step for the valsartan methyl ester intermediate. This results in the final product valsartan requiring multiple recrystallization to meet the pharmacopoeia standards in terms of chemical and optical purity. The operation is complex, the yield is low, and the cost is high. The medical-grade filtration, washing and drying three-in-one product developed by Wuxi Srui has solved the problems of low chemical and optical purity of valsartan produced by the existing process technology, which requires multiple recrystallization to meet the pharmacopoeia standards.
The valsartan cycination reaction solution is stirred and reacted into a salt in a medical-grade three-in-one process of filtration, washing and drying. Then, it is filtered, washed and dried to obtain the pure valsartan methyl ester alkali metal salt. The pure valsartan methyl ester alkali metal salt is then mixed with inorganic alkaline water solution II, heated for hydrolysis, followed by acidification, extraction, one-time recrystallization and drying to obtain valsartan.
The three-in-one process flow of filtration, washing and drying for the refined antihypertensive raw material drug valsartan of pharmaceutical grade
(1) Stirring reaction of crude valsartan
The cycination reaction solution for preparing valsartan methyl ester was mixed with inorganic alkaline water solution I and stirred for reaction.
(2) Preparation of crude valsartan:
After the reaction was completed, the water layer was cooled to 10℃ in layers, the pH was adjusted to 1 with hydrochloric acid, and the crude valsartan was obtained through filtration and washing.
(3) Refined dissolution of valsartan
Add the crude valsartan obtained to ethyl acetate and heat until completely dissolved.
(4) Primary refinement of valsartan
After cooling to room temperature, continue to cool to 10℃ and store for 3 hours. Filter and wash the filter cake with a small amount of cold ethyl acetate. Drying to obtain the primary crystalline product of valsartan;
(5) Secondary refining of valsartan
The primary crystallization product of valsartan, ethyl acetate, was heated to dissolve, stirred and cooled to room temperature, then filtered. The filter cake was washed with a small amount of ethyl acetate and dried to obtain the secondary crystallization product of valsartan, which is the refined valsartan.
Impurities in the refined valsartan were detected by HPLC. No relevant impurities were detected, no impurity K in valsartan was detected, and no chiral isomers were detected.
Compared with the traditional refining method, the three-in-one refining method of medical-grade filtration, washing and drying not only can reduce the isomer content of valsartan from 0.1 to 1.0% to N.D, and the chromatographic purity of the obtained valsartan is higher than 99.9%, but also all solvent residues can be dried to below the detection limit in a relatively short time. Its amplification process is stable, the process repeatability is strong, and the operation is simple. It has low costs and is highly suitable for industrial production.
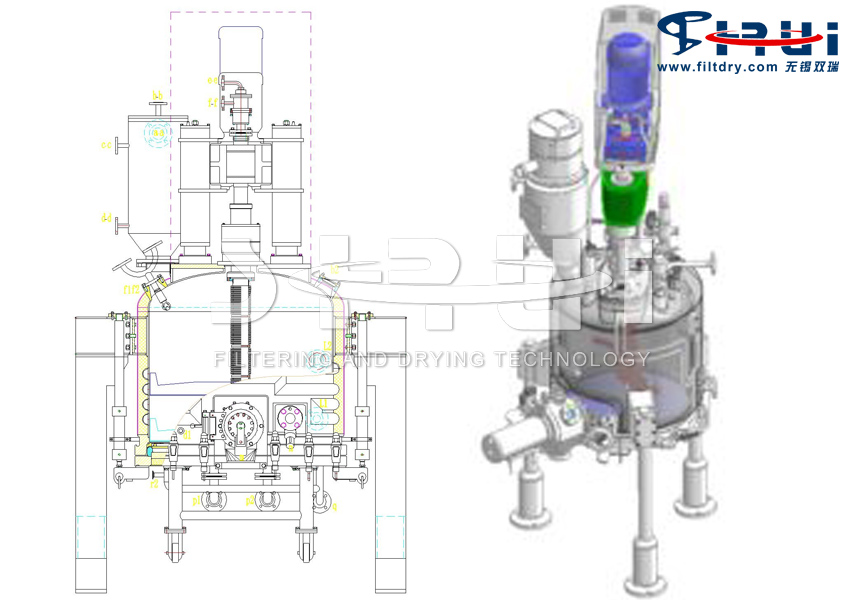
Photo of the medical-grade three-in-one equipment for filtration, washing and drying of the antihypertensive raw material drug valsartan















 备案号:
备案号: