Simvastatin is A commonly used lipid-lowering drug in clinical treatment at present. Simvastatin was developed by Merck of the United States. The basic pharmacological action of simvastatin is to limit the activity of hydroxymethylglutaryl-coA reductase in human liver cells through competitive inhibitors, thereby restricting the conversion of hydroxymethylglutaryl-CoA reductase to methyldihydroxyvaleric acid in liver cells. Ultimately, the goal of lowering cholesterol is achieved.
In clinical research, simvastatin can also play a certain role in preventing atherosclerosis and effectively avoid the recurrence of heart disease. Meanwhile, in the latest research, simvastatin, when used as a lipid-lowering drug, can also exert a certain degree of non-lipid-lowering effect. Firstly, simvastatin can promote the formation of new bone mass in the human body, thereby playing an auxiliary role in the clinical treatment of osteoporosis. Secondly, it can effectively reduce lipoprotein, lower blood pressure and has anti-tumor effects.
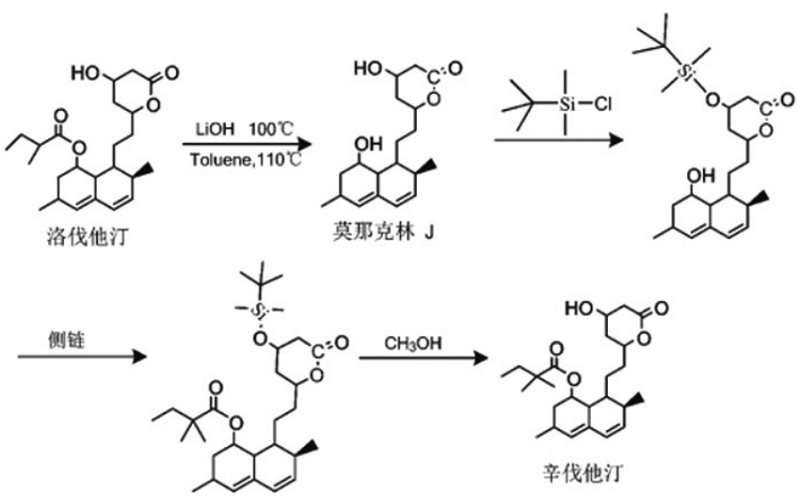
The current demand for improving the commonly used processes of high-purity simvastatin cardiovascular and cerebrovascular drugs
Indirect methylation, as a commonly used process for the synthesis of simvastatin at present, requires the use of a certain amount of organic solution, butyllithium. Butyllithium is a very active reagent. Under improper operation, it is very easy to cause serious explosion accidents, threatening the personal safety of production personnel. Meanwhile, the indirect methylation synthesis method of simvastatin can only be carried out under low-temperature conditions, and the production conditions are rather complex. This synthesis method still has problems such as multiple reaction steps, long reaction time required, low substrate conversion rate during the reaction process, a large number of types and quantities of by-products not needed for reaction production, and difficulty in purifying the finished product.
To meet the requirements of large-scale industrial production of simvastatin, the waste liquid produced by the new process is relatively easy to recover, will not cause serious environmental pollution, and the reaction conditions required during the reaction process are relatively mild. Therefore, the refining process of high-purity simvastatin cardiovascular and cerebrovascular drugs has been improved to a certain extent. After the reaction, water is no longer used for extraction. Instead, organic bases and the acid generated by the reaction are added to form complex salts, which are then precipitated in a solvent with lower polarity and filtered out. This avoids the decomposition of lovastatin protective substances upon contact with water, which could affect the subsequent reaction.

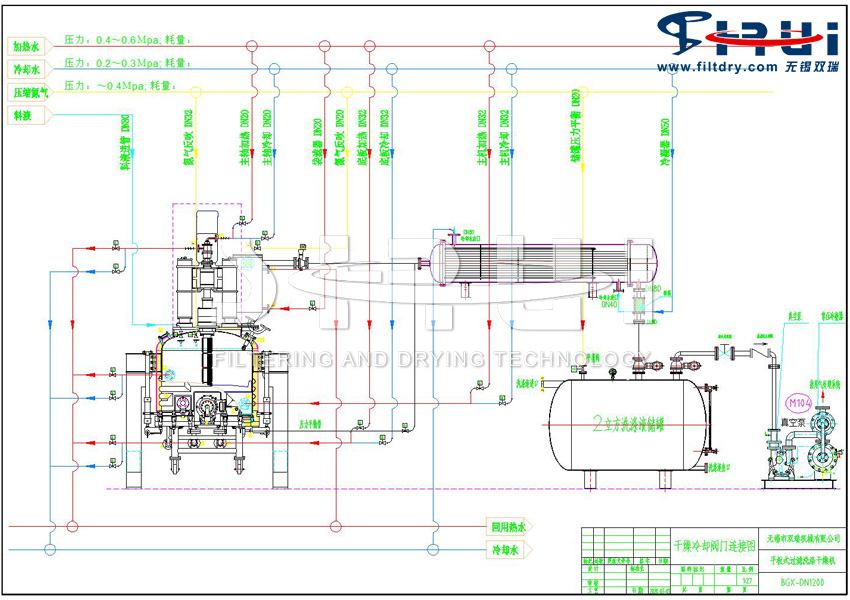
New Progress in the Refining Process of Pure simvastatin Cardiovascular and Cerebrovascular Drugs - Fully automatic Filtration, Washing and Drying Integrated Machine
Preparation of ammonium simvastatin salt
The lovastatin silanide was cooled down using a fully automatic filtration, washing and drying integrated machine. Then, a certain amount of lithium pyrrolidine was added and stirred. Finally, a certain amount of methyl iodide was added for methylation reaction. Make sure the reaction is complete, then add an appropriate amount of ether and water, adjust the acid and wash with water for extraction, and then evaporate the solvent dry.
Dissolve in methanol, stir and react at 25-30℃, and detect the end point of the reaction by HPLC. After the reaction is complete, ethyl acetate is added and extracted in layers. Wash the organic layer once again with saturated brine, add methanol, stir and drop ammonia water, cool to 5℃ and stir to crystallize. Simvastatin ammonium salt was obtained through filtration and drying.
(2) Preparation of simvastatin
In the fully automatic filtration, washing and drying integrated machine, the ammonium salt of simvastatin synthesized in the previous step is dispersed and suspended in the dichloromethane. Methanesulfonic acid is added at a temperature between 15℃ and 20℃. After ensuring the reaction is complete, solid sodium bicarbonate is added, activated carbon is filtered, and the dichloromethane is evaporated and dried. Then, ethanol is added and the temperature is raised to 55℃ to 60℃ for stirring and dissolution. Activated carbon is then added for decolorization and filtration. Between 45℃ and 50℃, water is dropped in, the temperature is cooled, crystallized, filtered, washed and dried to obtain the simvastatin product.

The fully automatic filtration, washing and drying integrated machine for the refining process of high-purity simvastatin cardiovascular and cerebrovascular drugs has fewer reaction steps, mild reaction conditions, key reactions taking place at room temperature, using less solvent, reducing environmental pollution, and the product quality is good, achieving good economic and environmental benefits. At the same time, the reaction yield is also relatively high and the quality is relatively stable. In the field of chemical synthesis, it is currently the most widely adopted refining and purification process route by domestic pharmaceutical enterprises.













 备案号:
备案号: