With the continuous increase in the state's support for new energy vehicles, the market share of new energy vehicles has been growing year by year. Lithium-ion batteries, as the main power and energy storage equipment for new energy vehicles, have also witnessed explosive development. The four key materials of lithium-ion batteries include the positive electrode, negative electrode, separator and electrolyte. Among them, the electrolyte plays a role in transferring charges between the positive and negative electrodes in the battery. Its quality directly affects the battery's cycle life, safety performance and energy density. Lithium hexafluorophosphate LiPF6, with its excellent ionic conductivity, electrochemical stability and the ability to form a stable passivation film on aluminum foil, has been widely used in lithium-ion batteries. However, LiPF6 has poor stability and is prone to decomposition reactions. It is also likely to explode at high temperatures, which is not conducive to storage and use.
Lithium bis (fluorosulfonyl) imide (abbreviated as LiFSI), as an important additive for lithium-ion battery electrolytes, features excellent electrochemical stability, good hydrolysis resistance and high electrical conductivity. It can be widely used in electrolytes, especially in power batteries, where it can improve the cycle performance and rate performance of power batteries. Lithium bis (fluorosulfonyl) imide is a new type of lithium electrolyte salt used in lithium battery electrolytes. It is environmentally friendly and has good safety performance, meeting the basic conditions for industrial application. Compared with the traditional lithium salt lithium hexafluorophosphate, lithium ions in lithium bis (fluorosulfonyl) imide are more prone to dissociation, thus having a higher electrical conductivity. The decomposition temperature of lithium bis (fluorosulfonyl) imide is above 200℃, and its thermal stability and safety performance are significantly better than those of lithium salt lithium hexafluorophosphate. In addition, it has unique effects in improving performance such as high-temperature storage and low-temperature discharge, and possesses excellent properties such as good compatibility with electrodes. Therefore, lithium bis (fluorosulfonyl) imide is an electrolyte with promising prospects in lithium-ion batteries.

At present, there are mainly two routes for the preparation technology of lithium bis (fluorosulfonyl) imide that have been publicly reported at home and abroad:
(1) The product is obtained by the double decomposition reaction of potassium bis (fluorosulfonyl) imide with organic lithium salts such as lithium tetrafluoroborate or lithium perchlorate in a non-protic organic solvent, generating lithium bis (fluorosulfonyl) imide and potassium tetrafluoroborate or potassium perchlorate. After filtration, separation, evaporation, concentration and crystallization, the product is obtained. Its drawbacks lie in: ① The double decomposition reaction is difficult to be complete, and the residual potassium ions in the product are relatively high; Lithium tetrafluoroborate and lithium perchlorate, which are used as reaction raw materials, have a high solubility in the reaction organic solvents and are difficult to separate. The resulting products often contain some unreacted or excessive raw materials, and the purity of the products is not high. ③ The prices of raw materials such as lithium tetrafluoroborate or lithium perchlorate and other organic lithium salts are relatively high, resulting in high manufacturing costs. Lithium perchlorate is an explosive compound that poses certain safety risks and is not suitable for industrial production.
(2) Bis (fluorosulfonyl) imide undergoes acid-base neutralization reactions with basic lithium salts such as lithium hydroxide or lithium carbonate in low-polarity organic solvents, generating lithium bis (fluorosulfonyl) imide and water. Then, by adding sulfoxide to react with water, hydrogen chloride and sulfur dioxide gas are produced to achieve the purpose of water removal. Filtration, solid-liquid separation, and further purification treatment are carried out to obtain the product. Its main drawbacks lie in: ① bis (fluorosulfonyl) imide is a protonic strong acid that reacts vigorously with bases, generating a large amount of heat. The reaction process requires low-temperature freezing control, which leads to high energy consumption. ② This reaction is essentially an acid-base neutralization reaction, simultaneously generating equal moles of water. The water removal reaction using dichloride is difficult to be thorough, with a long reaction time and a large amount of three wastes. Meanwhile, the water removal reaction process is prone to partial hydrolysis of the product, resulting in residual water, excessive chloride ions and sulfate ions in the product, making it difficult to obtain high-quality products.
The optimized purification process of lithium bis (fluorosulfonyl) imide by the conical filtration, washing and drying machine features mild reaction conditions, easy operation, and generates less waste, which is environmentally friendly. It can relatively conveniently obtain high-purity lithium bis (fluorosulfonyl) imide products with high technical indicators. The residual moisture, free acid, fluoride ions, chloride ions and other impurities in lithium bis (fluorosulfonyl) imide products can be controlled at a relatively low level, and it has a high reaction conversion rate and yield.
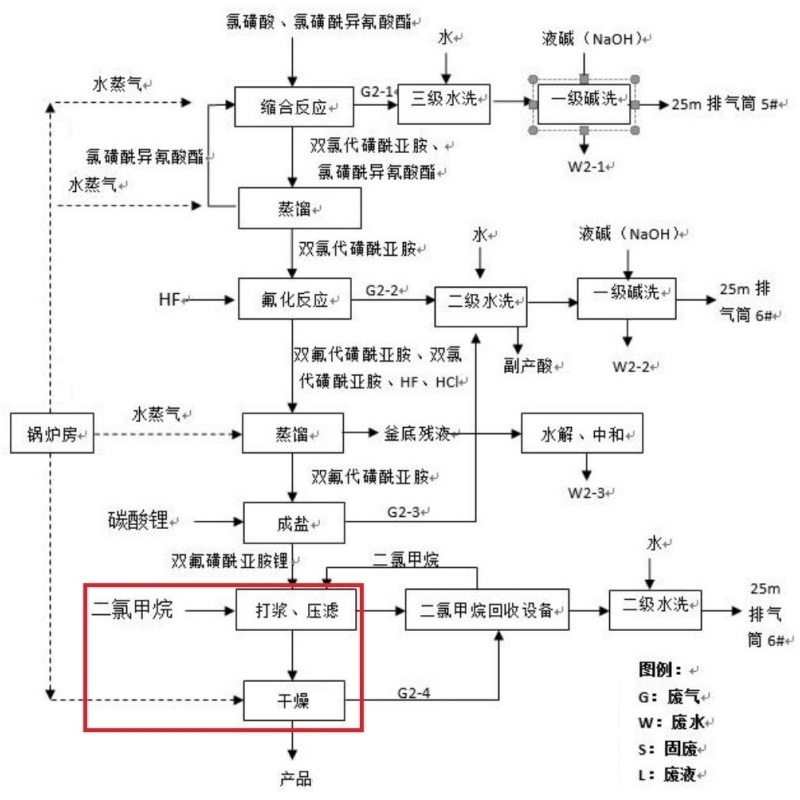
The process steps for purifying lithium bis (fluorosulfonyl) imide by conical filtration, washing and drying machine
The reaction liquid enters the concentration and crystallization reactor through pipelines, is heated to 25-30 ℃ for distillation, and then undergoes a first-stage cryogenic treatment of -15℃ brine and -50℃ to recover a certain amount of hydrogen fluoride for 2 to 3 hours. The temperature for the low-temperature crystallization process is controlled at -35℃ for 3 hours.
After crystallization, it enters the three-in-one filtration process. The mother liquor is distilled and then undergoes first-stage freezing brine at -15℃ and deep freezing at -50℃ to recover hydrogen fluoride. The solid is filtered by adding dichloromethane solvent in the three-in-one process for washing and filtration. The mother liquor of the filter is distilled and the dichloromethane is recovered through a first-stage cryogenic treatment of 15℃ chilled brine at +-50℃. The crude product was dried under reduced pressure to obtain lithium bis (fluorosulfonyl) imide.
(1) Under the condition of mixing and stirring anhydrous lithium halide salts with non-polar solvents, bisfluorosulfonyl imide is added dropwise, and the tail gas is absorbed by alkaline solution. After the addition is completed, continue the reaction at 0 to 60℃ for 1 to 12 hours, then cool to room temperature and add organic basic compounds to adjust the pH of the reaction solution to 5 to 8. Filtration is carried out, and the filter cake is rinsed with a non-polar solvent to obtain the crude product of lithium bis (fluorosulfonyl) imide.
(2) The preparation method of lithium bis (fluorosulfonyl) imide also includes dissolving the crude product of lithium bis (fluorosulfonyl) imide in a non-protic polar organic solvent, filtering to remove insoluble substances, concentrating the filtrate under reduced pressure, and then adding a poor solvent for crystallization. After filtration, washing and drying, high-purity lithium bis (fluorosulfonyl) imide products are obtained. The purity of this lithium bis (fluorosulfonyl) imide is ≥99.70%, the moisture content is ≤30ppm, the acidity (calculated as HF) is ≤50 PPM, the free chlorine content is ≤10ppm, and the impurity ions such as potassium and sodium in metals are all ≤5ppm.
(3) Aprotic polar organic solvents are ester compounds, ketone compounds, nitrile compounds or ether compounds. Selected from one or two of dimethyl carbonate, diethyl carbonate, methyl ethyl carbonate, propylene carbonate, methyl acetate, ethyl acetate, propyl acetate, isopropyl acetate, acetone, methyl ethyl ketone, methyl ethyl ketone, acetonitrile, propionitrile, ether, isopropyl ether, methyl tert-butyl ether, dimethyl glycol ether, diethyl glycol ether, 1, 4-dioxane, tetrahydrofuran or 2-methyltetrahydrofuran The mixture of the above.
(4) The addition amount of aprotic polar organic solvents is 0.8 to 5 times the mass multiple of the crude product of lithium bis (fluorosulfonyl) imide. Considering energy consumption, smooth filtration operation and other aspects comprehensively, the addition amount of the aprotic polar organic solvent is preferably 1 to 3 times the mass multiple of the crude product of lithium bis (fluorosulfonyl) imide.
(5) The non-polar solvent is a halogenated alkane, selected from one or a mixture of two or more of dichloromethane, chloroform, carbon tetrachloride, dichloroethane, trichloroethane, tetrachloroethane, dichloropropane, trichloropropane, and tetrachloropropane. Preferably, dichloromethane and dichloroethane, which have relatively lower toxicity, are preferred. More preferably, when dichloromethane is the non-polar solvent, the temperature for the holding reaction is controlled at the reflux temperature of 60℃.
(6) The addition amount of non-polar solvents is 1 to 5 times the mass multiple of bis (fluorosulfonyl) imide.
(7) Halogenated alkanes have good solubility in non-polar solvents, while the product lithium bis (fluorosulfonyl) imide is almost insoluble in this type of solvent. A good separation effect can be achieved through simple filtration.
(8) This process is an anhydrous system, thus fundamentally avoiding the hydrolysis of reaction raw materials and target products under heating and acidic conditions.
The working principle of the conical filtration washing and drying machine for purifying lithium bis (fluorosulfonyl) imide
(1) Pressure filtration or vacuum filtration
After the reaction is completed, open the lower filtrate outlet and introduce compressed air or nitrogen and other gases into the equipment to pressurize and filter the solid-liquid mixture that has undergone the reaction until the material inside the machine is completely dried. Vacuum can also be drawn at the filtrate outlet to perform vacuum filtration on the material until the filtrate is completely drained. During filtration, the hollow shaft and hollow screw ribbon can be rotated clockwise (viewed from the top down) to lift the material. At the same time, the hollow screw ribbon can limit the thickness of the filter cake layer while rotating, allowing the material to be filtered under the condition of a thin filter cake layer, thereby increasing the filtration rate. For materials that are easy to filter, there is no need to stir during filtration; just filter the materials dry directly.
(2) It can be washed and filtered multiple times
Some materials need to be washed multiple times after filtration. The reaction filtration and drying multi-functional machine is an ideal device for repeated and multiple washes of materials. After the material is pressed dry in the machine, washing liquid can be added to the top washing liquid inlet. Then, through the stirring of the hollow shaft and hollow screw ribbon, the filter cake is uniformly re-pulped. After the re-pulping is stirred evenly, the material is pressed dry again. If one wash is not enough, multiple washes can be carried out. At this time, just repeat the above steps several times. The operation is convenient and the labor intensity is extremely low.
(3) Vacuum drying
The filter cake that has been filtered or washed and dried remains in the machine. At this time, heat medium is introduced into the jacket of the cylinder, the jacket of the cone, the hollow shaft and the hollow screw ribbon to heat the material. Meanwhile, the hollow shaft and the hollow screw ribbon still rotate clockwise (viewed from the top) to lift the material upwards and stir it thoroughly. At the top of the equipment, vacuuming is carried out. Enable the materials to undergo vacuum and sealed drying inside the machine. Due to the fact that, in addition to jacket heating, the hollow shaft and hollow helical ribbon are also heated from the inside, and the drying is carried out under vacuum conditions, the drying efficiency is very high.
(4) Pulsating vacuum deep drying
The vacuum drying process uses a vacuum system to extract the inert gas inside the tank, reducing the number of gas molecules and isolating it from air. It is mainly suitable for materials that are prone to oxidation and deterioration, as well as for materials where solvents need to be recovered during evaporation and heat-sensitive materials. Under vacuum conditions, the chance of material contamination by bacteria can be reduced or the growth of certain bacteria can be inhibited. Therefore, it is particularly suitable for the drying of sterile materials.
There are several heating methods for the integrated filtration, washing and drying machine: the cylinder is equipped with half tubes or jackets, which are the main heating components provided; the heating device inside the chassis; and the hollow blades connected through the rotary joint at the shaft end. These heating structures can be used in combination or individually.
In the later stage of drying, the three-in-one equipment can first carry out vacuum drying for a certain period of time, then atmospheric pressure drying, and finally vacuum drying. This cycle repeats (i.e., using the pulsating vacuum drying method), which can improve the drying efficiency and shorten the drying time.
After the drying process is completed, open the quick-discharge port at the bottom of the equipment, and rotate the hollow shaft and hollow screw ribbon counterclockwise (viewed from the top down). The dried materials inside the machine will be automatically pushed towards the discharge port. At this point, the reaction, filtration, washing and drying processes of a batch of materials are completed.













 备案号:
备案号: