Due to the poor chemical stability of lithium hexafluorophosphate, it decomposes significantly when the temperature is above 55℃ or when LiPF6 comes into contact with protic solvents (such as water, alcohols, etc.), generating impurities such as phosphorus pentafluoride (PF5), phosphorus trifluoride (OPF3), and hydrogen fluoride (HF), which are highly destructive to the electrolyte and electrode materials. This is an important reason for the shortened service life of batteries and also the root cause of battery safety issues. Therefore, the development of conductive lithium salts with superior chemical and electrochemical stability has been a goal that both the academic and industrial communities have been tirelessly pursuing over the past two decades. Among them, the derivative of LiBF4, lithium difluoroxalate borate, due to its excellent chemical and electrochemical stability, significant passivation of Al current collectors at high potentials, and direct participation in the formation of the SEI film on the negative electrode surface, etc. It is regarded as one of the most promising new lithium salts to replace LiPF6, or can be used as an auxiliary salt in combination with LiPF6. Research shows that the chemical structure of LiDFOB lies between that of LiBOB and LiBF4, combining the advantages of lithium bis oxalate borate and lithium tetrafluoroborate. Its thermal decomposition temperature is as high as 240℃, and it has a relatively high solubility in carbonate solvents. Meanwhile, its relatively flexible chemical structure is conducive to reducing the viscosity of the electrolyte. Lithium difluoroxalate borate has good compatibility with graphite electrodes. The SEI film it participates in forming can maintain good stability at high temperatures. Therefore, lithium difluoroxalate borate simultaneously possesses the high-temperature performance of LiBOB and the low-temperature characteristics of LiBF4.

In the existing technology, lithium tetrafluoroborate and anhydrous oxalic acid are mostly reacted in organic solvents (such as dimethyl carbonate, etc.) to prepare lithium difluoroxalate borate, and a large amount of organic solvents are needed in the preparation process. Organic solvents are highly irritating and corrosive. When exposed to open flames, they can burn, posing a threat to the health of workers and the production environment. They also have high requirements for the corrosion resistance of equipment, resulting in high investment costs and being unfavorable for industrialization. Meanwhile, since lithium tetrafluoroborate and anhydrous oxalic acid react in organic solvents to form lithium difluoroxalate borate and hydrofluoric acid, which is a weak acid to a strong acid, the reaction is impossible to proceed. Therefore, catalysts or reaction AIDS are often required to be added. However, the use of catalysts is prone to introducing impurities into lithium difluoroxalate borate, resulting in a relatively low purity of lithium difluoroxalate borate and failing to meet the raw material requirements for lithium-ion battery electrolytes.
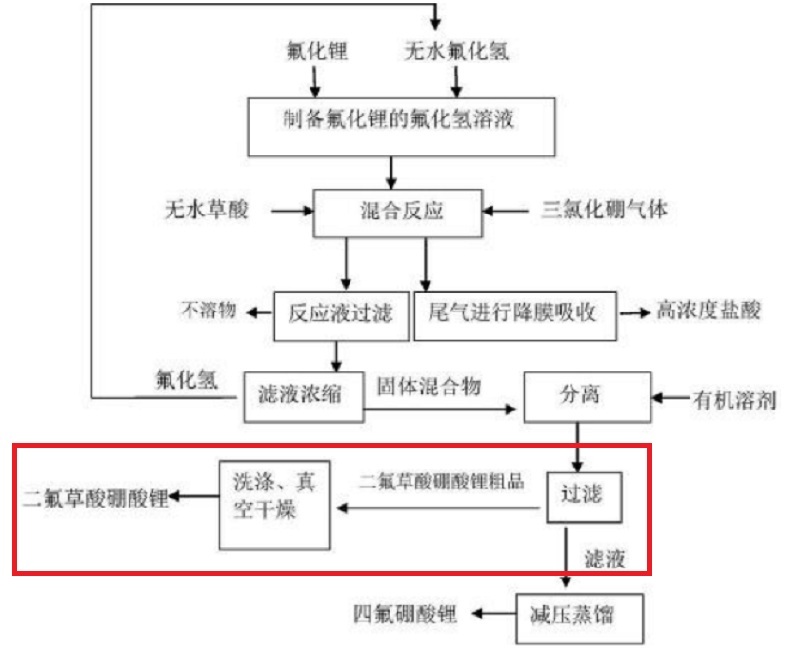
In the current process, when purifying lithium difluoroxalate borate powder, lithium difluoroxalate borate is first dissolved in a highly soluble solvent, then the solvent is mixed with a crystallization agent. Through solid-liquid separation, the crystallized solid substance is placed in a drying oven for drying, thereby obtaining lithium difluoroxalate borate powder and achieving the purification of lithium difluoroxalate borate. The existing purification devices have certain drawbacks when in use. They are not convenient for quantitative purification of the solid substances crystallized out, and the purification effect is poor, which brings certain adverse effects to people's usage process.
In response to the problems existing in the purification and drying technology field of lithium salts used as electrolytes in the lithium-ion battery industry, Wuxi Sunray has developed a conical drying system for purifying lithium difluoroxalate and borate (Patent No. : (202122464595.4) It adopts an automatic fully sealed multiple washing, nitrogen positive pressure filtration and pulse vacuum low-temperature drying process design. The operation is simple, the production cost is low, and it is economical and environmentally friendly. It can obtain high-purity lithium difluoroxalate borate, which meets the raw material requirements for lithium-ion battery electrolyte and is relatively suitable for industrial production.
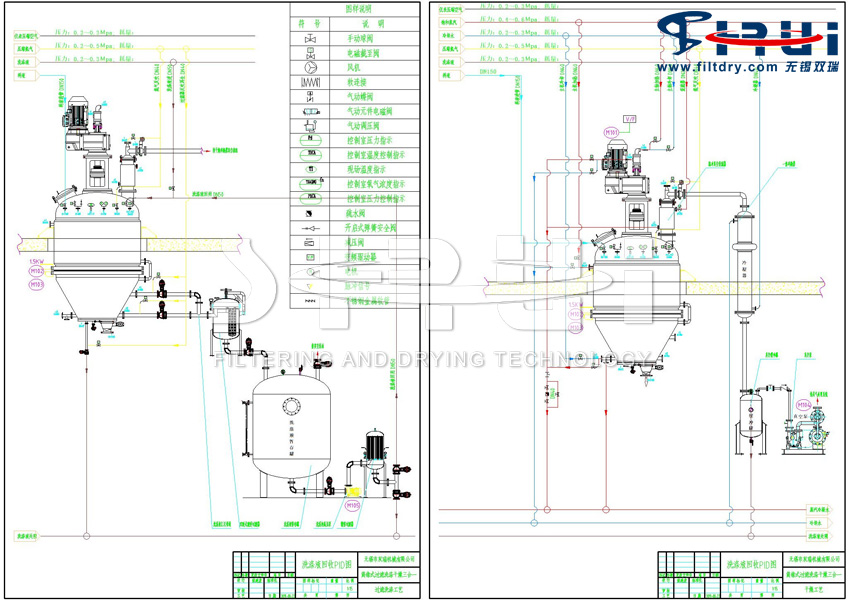
The lithium difluoroxalic acid borate conical drying system utilizes a hollow spiral stirring device to ensure uniform stirring of the material within the machine. It simultaneously heats or cools the material through the first heating jacket, the second heating jacket, the hollow shaft, and the hollow spiral blades, enabling the material to quickly meet the process requirements within the machine during the purification and drying stage.
During the filtration stage, this machine can make the material dry through pressure or vacuum filtration inside the machine, and add washing liquid to make the material undergo multiple automatic re-slurry washing inside the machine. After meeting the washing requirements, it can be pressed dry again. During the drying stage, the first heating jacket, the second heating jacket, the hollow shaft, and the hollow helical blades simultaneously heat the material and create a vacuum at the top, conducting vacuum sealed low-temperature drying of the material. After drying, the material is automatically sealed and discharged.














 备案号:
备案号: