Glyphosate acid or its salts are raw materials that can be used to prepare pesticides. Glyphosate is a commercially important and highly effective plant growth inhibitor. It can be used to suppress a variety of weeds and crops. This plant growth inhibitor can be applied to a very broad spectrum of perennial and annual grasses as well as the leaves of broad-leaved plants, thereby achieving the desired inhibitory effect. It has now become the herbicide with the largest sales volume in the world. In 2003, its sales reached 4.7 billion US dollars, accounting for 16% of the entire pesticide market.

There are various known methods for preparing glyphosate acid in terms of process. It mainly involves oxidizing bisphosphonate to glyphosate with a gas containing molecular oxygen as the oxidant in the presence of a catalyst composed of activated carbon. However, the reaction time for all of them is more than 12 hours, and the by-product formaldehyde generated cannot be removed in time. The environmental protection pressure remains significant. With the current process, the oxidation time of bisphosphonate is relatively long, all over 10 hours, and a large amount of by-products such as formaldehyde and formic acid are generated. As a result, the purity of the glyphosate obtained is not very high, and the highest yield is only about 90%. In conclusion, although the current technology can achieve the conversion of glyphosate and realize industrial production, there are still quite a few defects and deficiencies.
Analysis of Glyphosate Desalination and Purification Process
Cooling and crystallization
Reduce the temperature of the reaction vessel to 40℃ and then maintain it. After the heat preservation is completed, stop the air supply and add 10% ferrous sulfate solution drop by drop. A large amount of white sediment is formed upon cooling.
(2) Filtration and washing
The material liquid after cooling and crystallization was filtered and separated by a fully sealed two-in-one filter. The sediment was washed with water twice and then dried, and samples were taken for analysis. The filtered bisphosphonate mother liquor is partially or fully returned for the synthesis of bisphosphonate.
(3) Reuse of washing solution
The reaction residual liquid and washing water after separation and sedimentation are left to stand and stratified. The organic solvents and anthraquinones in the upper oil layer are recovered, while the water layer is discarded.
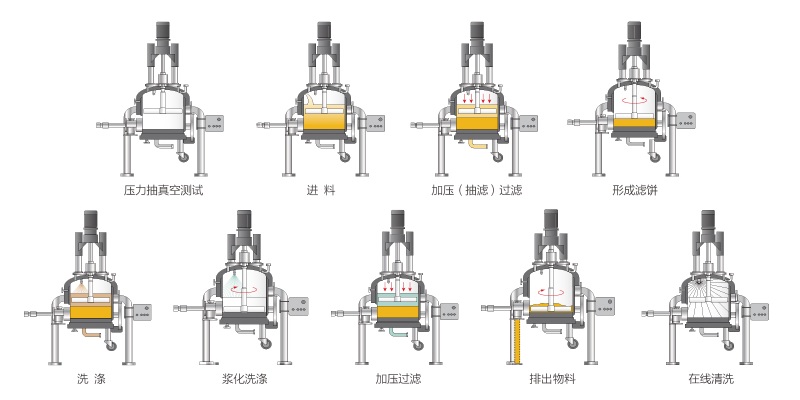
The fully sealed two-in-one filter enhances the purity of glyphosate. The purity of the glyphosate solid obtained is greater than 96%, and can reach up to 98% at the highest. It has the advantages of energy conservation and consumption reduction, shortened reaction time, increased reaction yield, reduced chemical reaction by-products, and no "three wastes". At the same time, it improves the raw material utilization rate of the entire glyphosate generation process, so the cost is lower.











 备案号:
备案号: