Glufosinate-ammonium was successfully developed by the German company Hoechst (which later became part of Bayer). It is an organophosphorus herbicide, a glutamine synthesis inhibitor, and a non-selective contact herbicide. At present, glufosinate-ammonium can be used in orchards, vineyards, non-cultivated land, potato fields and other environments to control annual and perennial dicotyledonous and grassy weeds, as well as perennial grassy weeds and sedges, with good effects.
Glufosinate-ammonium is a non-selective (non-selective) broad-spectrum contact herbicide and is one of the large-tonnage pesticide varieties in the world at present. Compared with glyphosate, glufosinate-ammonium has a better removal effect on some perennial noxious weeds and can also kill glyphosate-resistant weeds.Unlike glyphosate killing roots, glufosinate kills leaves first and can be carried out in the xylem of plants through transpiration, and its immediate effect is between paraquat and glyphosate. Many weeds are sensitive to glufosinate-ammonium and can be used as a substitute for glyphosate in areas where glyphosate resistance has developed.

Analysis of the purification production process technology of conventional glufosinate-ammonium
The crude product of glufosinate-ammonium contains a large amount of inorganic salt by-products such as ammonium chloride and generates viscous organic impurities. The conventional domestic purification process of glufosinate-ammonium is as follows: glufosinate-ammonium undergoes esterification reaction, hydrolysis reaction and neutralization reaction with ethylene oxide in sequence to obtain glufosinate-ammonium acid, and finally ammonia gas is passed to obtain high-purity glufosinate-ammonium. The route of this process is relatively long and the steps are rather complicated. Moreover, ethylene oxide is used in the step of removing hydrides. Although the purity of the glufosinate-ammonium salt obtained in the end is relatively high, the use of ethylene oxide has brought great limitations to this process. Ethylene oxide is a flammable and explosive gas and a toxic carcinogen. During its use, there are significant safety hazards. Moreover, due to its flammable and explosive nature, it is difficult to transport over long distances, thus having strong regional characteristics, which brings inconvenience to production and also increases costs.
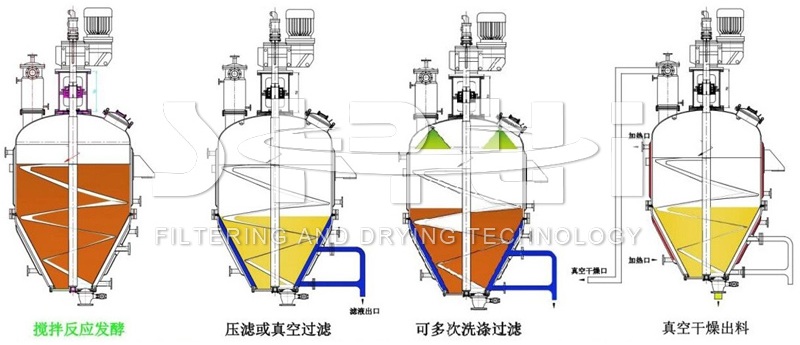
The fully automatic filtration, washing and drying three-in-one glufosinate-ammonium purification production process flow
The fully automatic filtration, washing and drying three-in-one glufosinate-ammonium purification production process is a green, environmentally friendly and low-cost purification method for glufosinate-ammonium, and the purity and yield of the obtained glufosinate-ammonium are high. The specific process is as follows:
(1) Fully automatic reaction filtration process
Dissolve the crude glufosinate-ammonium hydrochloride in a solvent, then add amino acids and alkali, adjust the pH to 7 to 10, react for 0.5 to 1.5 hours, distill under reduced pressure to remove part of the solvent, and then filter to remove ammonium chloride and other insoluble substances.
(2) Fully automatic one-time filtration and washing process
Add acid to the reaction solution in step (1) for reaction, adjust the pH to 1 to 5, and then filter and dry to obtain glufosinate-ammonium acid.
(3) Fully automatic secondary filtration, washing and drying process
The glufosinate-ammonium acid obtained in step (2) is dissolved in a solvent, and then ammonia gas is introduced for salt formation reaction to precipitate glufosinate-ammonium solid. After filtration and drying, high-purity glufosinate-ammonium is obtained.

The fully automatic three-in-one purification production process of filtration, washing and drying of glufosinate-ammonium avoids the use of difficult-to-remove aliphatic amines and highly dangerous ethylene oxide and propylene oxide. The steps are simple, and the obtained glufosinate-ammonium has a low content of inorganic salts, high purity, high yield and high safety. The process uses water or alcohol as the solvent and neutralizes with ammonia water and inorganic alkali, which conforms to the principles of environmental protection and economy. It has a very good prospect for industrialization.














 备案号:
备案号: